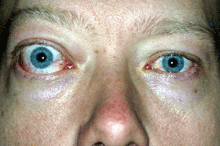
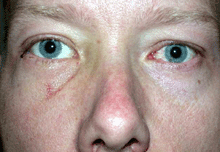
In TED optic neuropathy, the optic nerve becomes compressed by the enlarged muscles at the narrow orbital apex where the nerve exits the orbit to enter the brain cavity. This interferes with flow of information through the optic nerve connecting the eyes to the brain.
The patient symptoms may include central visual blurring, loss of peripheral vision or colour desaturation. The progress and duration of symptoms is also noted.
Objective measures of optic neuropathy include a loss in central visual acuity and colour vision, an afferent pupil defect, and infrequently congestion or pallor of the optic nerve.
Ancillary testing includes coronal CT or MR Scans to confirm crowding of the orbital apex, standardized visual fields, and rarely VEP or optic nerve head photos. As a summary grade, VISA lists optic neuropathy as present or absent since most clinicians would attempt some treatment for this condition if present. The severity of the neuropathy is reflected in the individual measurements of central and colour vision.
Fortunately, the optic neuropathy in TED is often partially or completely reversible. The common treatment for TED optic neuropathy initially is high dose corticosteroids either by intravenous route (1 gram Methyl-prednisolone over 30 minutes on alternate days for 3 treatments) or oral route (100 mg prednisone daily on a tapering dose) with an expectation that vision should improve within days of therapy. In most cases, this treatment will cause incomplete or only temporary visual improvement so that surgical decompression of the medial wall is required (through a Lynch, transcaruncular or endoscopic transethmoidal approach) or through a lateral approach (thinning the outer boney wall of the orbit near the apex) in order to relieve pressure more permanently on the optic nerve at the crowded orbital apex.
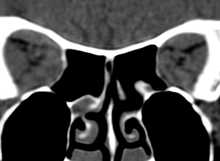
Coronal CT showing crowded orbital apex with compression of optic nerves
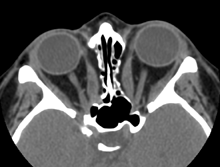
Axial CT Scan with medial wall de-compressions and freed optic nerves
Because the disease is usually in the active or progressive phase when optic neuropathy develops, some clinicians may arrange additional radiotherapy subsequent to the decompression to prevent further enlargement of the muscles and recurrence of neuropathy. Radiotherapy is administered using a lateral port focused behind the globe to minimize the risk of retinal or lens exposure; it is divided into 10 fractions of 200 rads over two weeks [23] and is contraindicated in diabetics because of the risk of inciting or aggravating retinopathy. [24] Although radiotherapy remains controversial, it is still widely used and many clinicians believe it is beneficial for certain aspects of TED, including optic neuropathy and significant inflammation. [25]
Success of therapy for TED optic neuropathy from both a clinical or research standpoint is based on specific improved measurements for central vision, colour vision and visual fields.

Symptoms of ocular and periocular soft tissue inflammation include orbital aching at rest or with movement, and eyelid or conjunctival swelling and redness.
The Clinical Activity Score (CAS) described and validated by Mourits and the Amsterdam Orbitopathy group assigns one point for each of the following: orbital pain at rest, orbital pain with movement, chemosis, caruncular edema, eyelid edema, conjunctival injection and eyelid injection. [21]
The VISA Inflammatory Score modifies the CAS slightly by widening the grade for chemosis and lid edema from 0-2. Chemosis (swelling of the conjunctiva) is graded as 1 if the conjunctiva lies behind the grey line of the lid and as 2 if it extends anterior to the grey line:
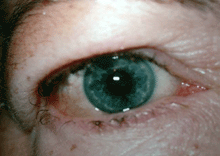
Grade 1 Edema (swollen without a roll)
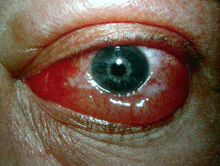
Grade 1 edema (right eye), grade 2 edema with festoon (left eye)
Lid edema is graded as 1 if it is present but not causing overhanging of the tissues, and as 2 if it causes a roll in the lid skin including festoons in the lower lid. The worst scores from any of the four eyelids are recorded in the inflammatory score table on the far right section of the table.
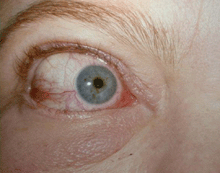
Grade 1 Edema (swollen without a roll)
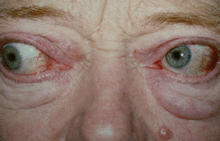
Grade 1 edema (right eye), grade 2 edema with festoon (left eye)
The pain score is based on the patient’s report of deep orbit discomfort rather than ocular surface irritation (0 = no pain, 1 = pain with movement, 2 = pain at rest). The additional grading scores for chemosis and lid edema allow for documentation of more subtle changes in inflammatory features between visits. An additional point is assigned for diurnal variation of symptoms, to reflect the variability in congestion typically seen during the active phase.
Treatment of active inflammation in TED depends on its inflammatory score and evidence of progression. If the score is less than 4 out of 10, and there is no deterioration based on history or sequential clinical examination, conservative management is offered with reassurance, cool compresses, nocturnal head elevation, and non-steroidal anti-inflammatories.
In general, if the inflammatory grade is 5 or more, or if there is subjective or objective evidence of progression in the inflammation, more aggressive therapy should be considered, including oral or intravenous corticosteroids, radiotherapy, and in refractory cases, immunosuppressive agents.
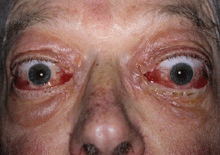
Pre-steroid treatment: VISA inflammatory score = 9/10
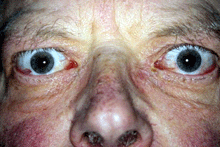
Post IV GC
Combination therapy is receiving increasing attention in severe, progressive cases [26, 27] and interest is also turning to new immunomodulatory agents, such as anti-tumor necrosis factor agents (etanercept, infliximab) or B-lymphocyte directed therapy (rituximab). [28] The hope from greater understanding of the immunogenic mechanisms in this disease is to identify markers of progressive, more severe disease so that earlier treatment and more specific immunotherapy may be developed.

The symptoms for strabismus include a progression from no diplopia (double vision), diplopia with horizontal or vertical gaze, intermittent diplopia in straight gaze, and constant diplopia in straight gaze.
Ocular ductions can be graded from 0o to 45o in four directions using the Hirschberg principle: the patient is asked to look as far as possible up, down, right and left while the observer points a bright light at the eyes and studies the light reflex on the surface of the eye. If the light reflex hits the edge of the pupil, the eye has moved 15o, between the pupil edge and the limbus, 30o and at the limbus, 45o.
Strabismus can be measured objectively by prism cover testing in different gaze directions.
Ancillary testing includes using the Goldmann perimeter to quantify ocular ductions in four directions. The patient keeps both eyes open and follows the V4e light target, tapping a coin when the image becomes double.
Management of strabismus depends on whether the orbitopathy is inflamed (measured in the previous section) or if there is evidence of progression in symptoms and signs. If inflammation is present (figure 4a below), this is managed first, either with conservative treatment or with anti-inflammatories or radiotherapy. During this stage, the strabismus can be managed with patching one eye or with Fresnel prisms. Once the inflammatory score has dropped to zero and there is no evidence of progression, management of strabismus might include prisms or surgical alignment (figure 4b).
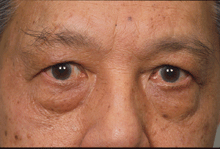
Figure 4a: Active inflammation with progressive strabismus. At this stage, immune modulatory medicines or radiotherapy may control progression and patching is offered to prevent double vision.
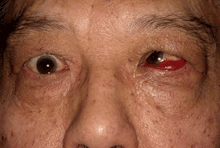
Figure 4b: Once the inflammation controlled and the strabismus measurements are stable, prisms may be offered or alignment surgery with adjustable sutures can be performed.

Symptoms in this category include appearance concerns such as bulging of the eyes, eyelid retraction and fat pockets, as well as exposure complaints of foreign body sensation, glare, dryness or secondary tearing.
Objective measures of appearance change include eyelid retraction (measured in millimeters), proptosis (measured with the Hertel exophthalmometer), and documentation of redundant skin and fat prolapse. Measures of exposure include corneal staining or ulceration.
Photographs can document the appearance changes.
Management of appearance and exposure changes depend on the inflammatory stage of the disease. During the inflammatory phase (documented progression in any of the parameters or an inflammatory score > 5), lubricant drops and ointments can relieve ocular irritation.
Rarely a tarsorrhaphy (temporarily suturing the upper and lower lids) or emergency orbital decompression may be required for severe exposure or corneal ulceration.
Once the inflammatory phase has settled, management for proptosis might include orbital decompression and for eyelid retraction may include upper lid lowering from an anterior or posterior approach or lower lid elevation.31 These surgical measures often relieve many of the exposure complaints.