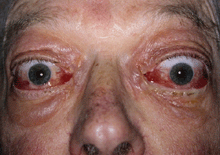
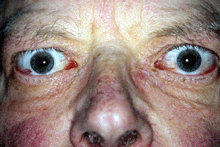
These remain the first choice for active TED with significant congestive or inflammatory signs. They may reverse DON, although this may be incomplete or temporary so that adjunctive radiotherapy or surgical decompression may be necessary. They also are recommended preventively to avoid exacerbation of active TED at time of radioactive iodine thyroid ablation.
Standard oral therapy is daily prednisone 0.5–1.0 mg/kg daily for 6 weeks, but this route has side effects in 50% of cases, including Cushing’s syndrome, diabetes mellitus, insomnia, mood disturbances, gastric ulcers, osteopenia, necrosis of the femoral head, and susceptibility to infections.
Intravenous GC therapy reduces the incidence of side effects and allows a longer duration of therapy. A popular protocol is IV methylprednisolone 500 mg weekly for 6 weeks, reducing to 250 mg weekly for 6 weeks. This author prefers to titrate the dose or prolong the intervals depending on clinical response. Liver failure and rare cases of death have been reported in cumulative doses over 8 g of methylprednisolone.
Depot GC injection into the inferolateral orbital fat pocket is beneficial for focal orbital congestion but carries a small risk of intravascular emboli with visual loss.